By Sneha S K and Sahil Pandey
Dec 31 (Reuters) - The U.S. Food and Drug Administration has declined to approve Corcept Therapeutics' drug for the treatment of a rare hormonal disorder, the company said on Wednesday.
Shares of the drugmaker were down 48% at $36.41.
The company said the FDA could not arrive at a favorable benefit-risk assessment for the hormone-blocking oral treatment, known as relacorilant, without Corcept providing additional evidence of effectiveness.
The company was seeking approval for relacorilant as a treatment for patients with hypertension secondary to hypercortisolism.
"FDA's request for additional data may require additional trials, significantly dimming Corcept's outlook in Cushings," said Truist analyst Joon Lee.
Hypercortisolism, also known as Cushing's syndrome, occurs when the body is exposed to high cortisol activity.
Corcept had submitted trial data that showed that relacorilant made improvements in a wide array of hypercortisolism's signs and symptoms.
"We will meet with the FDA as soon as possible to discuss the best path forward," said Joseph Belanoff, Corcept's CEO.
Main symptoms of hypercortisolism include a fatty hump between the shoulders, a rounded face, and pink or purple stretch marks on the skin. People with Cushing's also experience diabetes, high blood pressure, muscle weakness and immune suppression.
Relacorilant is a selective cortisol modulator designed to block the effects of cortisol, while avoiding certain off‑target hormonal effects.
"Given the company had opportunities to address FDA's concerns during mid and late-stage reviews, it's unclear if any further dialogue can resolve the review issues without additional trials," Lee added.
Corcept is also studying the drug in a variety of serious disorders including ovarian and prostate cancer. Its other drug known as Korlym is approved to treat high blood sugar caused by hypercortisolism in adults with endogenous Cushing's syndrome.
Other approved treatments for Cushing's syndrome include Isturisa by Recordati and Xeris Biopharma's Recorlev.
(Reporting by Sahil Pandey and Sneha S K in Bengaluru; Editing by Shailesh Kuber)
LATEST POSTS
- 1
 Blood pressure drug recalled for possible cross-contamination
Blood pressure drug recalled for possible cross-contamination - 2
 The pace of hiring just fell to the lowest since 2011, outside of the pandemic
The pace of hiring just fell to the lowest since 2011, outside of the pandemic - 3
 Police arrest 18 as anti-war protests spread across Tel Aviv, Haifa, Jerusalem
Police arrest 18 as anti-war protests spread across Tel Aviv, Haifa, Jerusalem - 4
 IDF Givati Brigade soldier arrested, charged in suspected Iran espionage
IDF Givati Brigade soldier arrested, charged in suspected Iran espionage - 5
 Oldest sequenced RNA reveals details about a mammoth’s final moments 40,000 years
Oldest sequenced RNA reveals details about a mammoth’s final moments 40,000 years
 People are getting their news from AI – and it’s altering their views
People are getting their news from AI – and it’s altering their views Luigi Mangione‘s lawyers say Bondi’s death penalty decision was tainted by conflict of interest
Luigi Mangione‘s lawyers say Bondi’s death penalty decision was tainted by conflict of interest Ukraine: Russians abduct 50 Ukrainians from border village in Sumy
Ukraine: Russians abduct 50 Ukrainians from border village in Sumy Don't miss Jupiter shining close to the waning gibbous moon on Dec. 7
Don't miss Jupiter shining close to the waning gibbous moon on Dec. 7 Figure out How to Augment Eco-friendliness in Your Volvo XC40
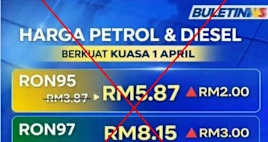
Figure out How to Augment Eco-friendliness in Your Volvo XC40 False fuel prices in fabricated graphics circulate in Malaysia as Iran war continues
False fuel prices in fabricated graphics circulate in Malaysia as Iran war continues 10 Activities to Lift Your Consume and Bust Your Stomach
10 Activities to Lift Your Consume and Bust Your Stomach The Best 15 Applications for Efficiency and Association
The Best 15 Applications for Efficiency and Association 5 Different ways Macintosh is Prepared to Overwhelm Gaming, Even Against Windows
5 Different ways Macintosh is Prepared to Overwhelm Gaming, Even Against Windows